Understanding and Mitigating Postprandial Somnolence ('Food Coma')
Introduction
The ubiquitous experience of postprandial somnolence, commonly referred to as 'food coma,' presents a significant yet often overlooked aspect of human physiology and daily life. Following a meal, a substantial portion of the population reports feelings of drowsiness, lethargy, and a reduced capacity for cognitive tasks. While often dismissed as a benign consequence of overindulgence, the pervasiveness of this phenomenon in modern society warrants a deeper investigation into its underlying causes and potential implications. Understanding the factors contributing to post-meal sleepiness is crucial for optimizing well-being, productivity, and potentially identifying early indicators of underlying health issues. This research endeavors to move beyond anecdotal observations to provide a scientifically grounded examination of food coma, addressing the growing need for clarity and actionable insights in this area.
This report specifically addresses the limitations in current understanding regarding the complex interplay of physiological responses, dietary habits, and the experience of postprandial somnolence. Existing explanations often oversimplify the phenomenon, failing to adequately account for the diverse range of individual experiences or the influence of contemporary dietary patterns characterized by processed foods and high-glycemic loads. The core research questions guiding this investigation are: What are the primary physiological mechanisms that induce drowsiness after eating? How do modern dietary choices and food composition exacerbate or mitigate these effects? And what evidence-based strategies can be employed to effectively reduce or eliminate this post-meal fatigue? By seeking answers to these questions, this research aims to bridge the gap between common experience and scientific understanding, offering a more comprehensive perspective on food coma.
The scope of this research is structured to systematically explore the multifaceted nature of postprandial somnolence. It will begin by dissecting the intricate physiological pathways involved, examining the roles of key hormones, neurotransmitters, and macronutrient metabolism in regulating alertness and sleepiness post-ingestion. Subsequently, the report will critically analyze the impact of modern dietary trends, focusing on the contribution of processed foods, refined carbohydrates, and meal composition to the intensity and frequency of food coma. Finally, the research will synthesize these findings to propose practical, health-conscious strategies for mitigation, considering both dietary adjustments and lifestyle modifications, and outlining when professional medical consultation is advisable. The logical progression from fundamental biological processes to applied solutions ensures a holistic and actionable outcome.
To facilitate a clear and structured understanding of the research findings, this report is organized into four distinct sections. The initial section provides this comprehensive introduction, setting the context and outlining the research objectives. The subsequent sections delve into the core research areas: the physiological mechanisms, the influence of dietary factors, and health implications alongside mitigation strategies. Each section builds upon the preceding one, creating a cohesive narrative that guides the reader through the complexities of postprandial somnolence. This approach is designed to equip readers with a thorough understanding of the phenomenon and practical recommendations for managing it.
1. Physiological Mechanisms of Postprandial Somnolence
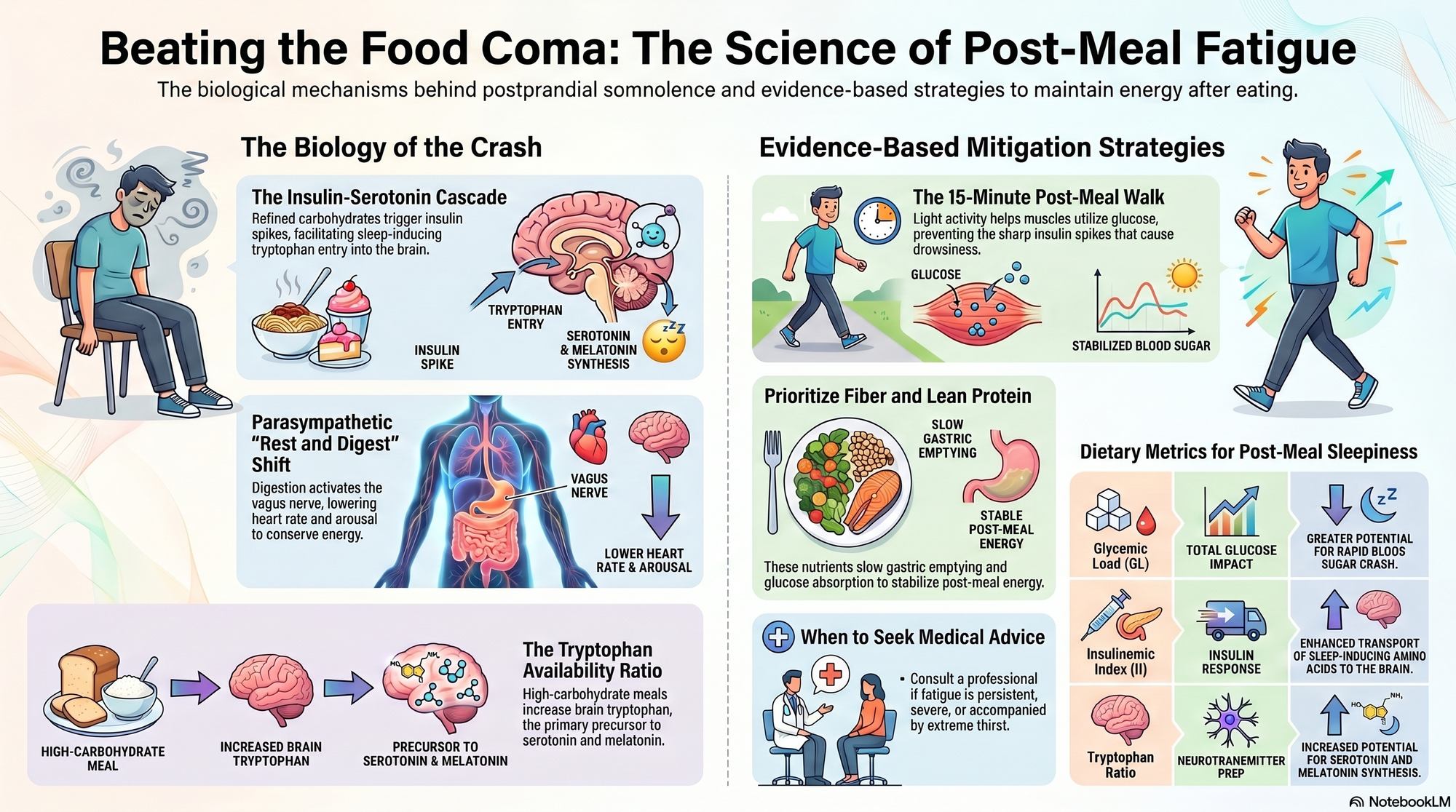
Postprandial somnolence, commonly referred to as 'food coma,' is a transient state of drowsiness, reduced alertness, and a desire for rest experienced after consuming a meal. This phenomenon is not merely a subjective feeling but is underpinned by a complex interplay of physiological processes involving macronutrient digestion, hormonal signaling, and neural pathways that integrate digestive feedback with the body's sleep-wake regulatory systems. Understanding these mechanisms is crucial for differentiating normal physiological responses from potential underlying health issues and for developing effective management strategies. The body's shift towards energy storage and restorative processes post-meal, mediated by specific hormonal surges and neural cascades, promotes a state of reduced alertness, often characterized by increased parasympathetic nervous system activity.
1.1 Macronutrient Influence on Somnolence
The composition of a meal, specifically its macronutrient profile (carbohydrates, fats, and proteins), significantly influences the magnitude and nature of postprandial physiological responses that can lead to somnolence. Different macronutrients are processed via distinct metabolic pathways, eliciting varied hormonal and neural signals.
Carbohydrates are a primary driver of rapid postprandial somnolence, particularly those with a high glycemic index (GI) and glycemic load (GL). High GI/GL meals lead to a swift absorption of glucose into the bloodstream, resulting in a pronounced postprandial hyperglycemia. This surge in blood glucose triggers a robust insulin response from the pancreas. Insulin's primary role is to facilitate glucose uptake by peripheral tissues (muscle and adipose tissue) and inhibit hepatic glucose production, thereby lowering blood glucose levels. However, insulin also has indirect effects on neurotransmitter synthesis. It promotes the uptake of most large neutral amino acids (LNAAs) into muscle cells, but tryptophan, a crucial precursor for serotonin and melatonin, competes less effectively for this uptake. Consequently, the plasma ratio of free tryptophan to other competing LNAAs increases, enhancing tryptophan's availability to cross the blood-brain barrier. This increased central nervous system (CNS) tryptophan availability is hypothesized to drive enhanced synthesis and release of serotonin, a neurotransmitter associated with mood regulation, reduced arousal, and increased sleep propensity [1].
Fats are digested more slowly than carbohydrates, leading to a more gradual release of energy and a less pronounced, prolonged postprandial glucose and insulin response. While high-fat meals do not typically induce the rapid somnolence associated with high-carbohydrate meals, the digestive process itself is metabolically demanding and can contribute to a general feeling of lethargy over a longer period. Furthermore, the presence of fats in the small intestine stimulates the release of cholecystokinin (CCK), a satiety hormone. CCK plays a role in signaling fullness to the brain and has been implicated in influencing sleep-wake cycles, though its direct contribution to immediate postprandial somnolence is complex and potentially modulated by other factors [1]. The prolonged digestion time of fats also means that the body remains in a state of active digestion for an extended duration, which can favor parasympathetic nervous system activity.
Proteins also stimulate digestive processes and hormonal responses. Like carbohydrates, proteins are composed of amino acids, including tryptophan. However, the impact of dietary protein on brain tryptophan levels is more nuanced. Proteins are typically digested into a mixture of amino acids, and the presence of other LNAAs from protein digestion can compete with tryptophan for transport across the blood-brain barrier. Therefore, while protein intake contributes to satiety and can influence hormonal profiles, its effect on brain serotonin synthesis is generally considered less potent than that of high-carbohydrate meals, especially when consumed in isolation or as part of a balanced meal where other amino acids compete for transport [1]. The specific amino acid profile of a protein and its interaction with other meal components can modulate these effects.
Collectively, the macronutrient composition dictates the overall metabolic and hormonal milieu post-meal. Meals high in refined carbohydrates and sugars tend to promote a more significant insulin surge, leading to a greater relative increase in brain tryptophan availability and thus a higher propensity for postprandial somnolence. Conversely, meals rich in fiber, protein, and healthy fats generally lead to a slower, more sustained release of energy and hormonal signals, potentially mitigating acute drowsiness.
1.2 Hormonal and Neurotransmitter Pathways
The regulation of alertness and sleepiness following a meal is heavily influenced by a cascade of hormonal and neurotransmitter signals originating from the digestive tract and acting on the central nervous system. Key players in this process include insulin, serotonin, melatonin, and gut-derived peptides like CCK.
Insulin, as discussed, is a critical mediator. Released in response to elevated blood glucose, its indirect effect on increasing brain tryptophan availability is a cornerstone of the carbohydrate-somnolence hypothesis [1]. The magnitude and duration of the insulin spike, influenced by meal composition and individual insulin sensitivity, directly correlate with the potential for this indirect neurotransmitter modulation. Quantitative metrics like the Tryptophan Availability Ratio (the ratio of free plasma tryptophan to the sum of other competing LNAAs) serve as proxies for brain tryptophan uptake. A higher ratio, often observed after high-carbohydrate meals due to insulin's action, suggests enhanced potential for serotonin synthesis [2, 3].
Serotonin is a key neurotransmitter synthesized from tryptophan. Within the brain, serotonin plays a multifaceted role, influencing mood, appetite, and crucially, sleep-wake cycles. Elevated serotonin levels are associated with reduced alertness and increased sleep propensity. The postprandial increase in brain tryptophan availability, facilitated by insulin, can lead to a greater flux of tryptophan into serotonergic neurons, thereby enhancing serotonin synthesis and release [1]. While direct measurement of serotonin synthesis rates in the human brain postprandially is challenging, the tryptophan availability ratio serves as an important indirect indicator [2].
Melatonin, another hormone derived from tryptophan, is primarily known for its role in regulating circadian rhythms and promoting sleep. While the direct link between acute postprandial somnolence and a surge in melatonin is less established than that of serotonin, tryptophan is its precursor. It is plausible that sustained increases in tryptophan availability post-meal could indirectly influence melatonin synthesis over a longer timeframe, contributing to a general shift towards a sleep-promoting state [1]. Further research is needed to clarify the direct contribution of postprandial melatonin fluctuations to immediate drowsiness.
Cholecystokinin (CCK) is a peptide hormone released by the duodenum and jejunum in response to the presence of fats and proteins. CCK plays a significant role in promoting satiety by slowing gastric emptying and signaling to the brain via the vagus nerve. While its primary function is related to appetite regulation, CCK signaling in the brain has also been implicated in sleep regulation. High postprandial CCK levels may contribute to feelings of fullness and potentially induce sleepiness, though its specific role in acute food coma is complex and may be modulated by other satiety hormones like ghrelin and leptin [1, 2].
Other gut hormones, such as Ghrelin (the 'hunger hormone') and Leptin (the 'satiety hormone'), also participate in the intricate hormonal dialogue following a meal. Postprandial suppression of ghrelin and increase in leptin signal satiety and energy availability, which can indirectly influence arousal states and contribute to a general feeling of reduced alertness over time [2]. The interplay between these various hormonal signals creates a complex regulatory network that influences both digestive processes and the regulation of sleep and wakefulness.
1.3 Neural Signaling and Digestive System Integration
The digestive system is richly innervated and communicates extensively with the central nervous system (CNS) through intricate neural pathways. This gut-brain axis is fundamental to integrating digestive status with overall physiological states, including alertness and sleepiness.
The vagus nerve is a primary conduit for this communication. As the longest cranial nerve, it forms a bidirectional communication highway between the brainstem and numerous visceral organs, including the stomach, intestines, and liver. Afferent fibers of the vagus nerve transmit sensory information from the gut to the CNS, signaling nutrient presence, stretch, and the release of various digestive hormones. For instance, signals related to gut distension and the presence of specific nutrients can activate vagal pathways that project to brainstem nuclei and subsequently influence hypothalamic centers involved in arousal and sleep regulation. This vagal signaling contributes to the feeling of satiety and can also promote a shift towards parasympathetic dominance, a state conducive to rest and digestion [1].
The hypothalamus, a small but vital region in the brain, acts as a central integrator of energy balance, appetite, and sleep-wake regulation. It receives and processes signals from the digestive system via hormonal and neural pathways, as well as from the bloodstream (e.g., glucose, leptin levels). The hypothalamus contains nuclei that orchestrate the body's response to feeding, including modulating neurotransmitter systems involved in arousal (e.g., orexin, serotonin, norepinephrine) and sleep promotion (e.g., GABAergic neurons). Following a meal, the hypothalamus may downregulate arousal-promoting pathways and upregulate sleep-promoting pathways in response to the integrated signals of nutrient availability and satiety, contributing to postprandial somnolence.
The concept of parasympathetic nervous system activation is central to understanding the physiological state post-meal. The parasympathetic nervous system is responsible for the 'rest and digest' functions, promoting energy conservation and restorative processes. In contrast, the sympathetic nervous system is associated with the 'fight or flight' response, characterized by increased alertness and energy mobilization. After a meal, the body naturally shifts towards parasympathetic dominance. This shift is characterized by a decrease in heart rate, increased gastrointestinal motility, and a general reduction in overall arousal, all of which contribute to the feeling of drowsiness and the desire to rest [1]. This activation is a normal physiological response aimed at facilitating digestion and nutrient assimilation.
1.4 Quantitative Modeling and Assessment Metrics
To rigorously investigate and quantify the physiological mechanisms underlying postprandial somnolence, researchers employ a range of quantitative models and assessment metrics. These tools allow for the precise measurement of metabolic and hormonal responses and their correlation with subjective and objective measures of alertness.
Glucose-Insulin Dynamics are frequently assessed using metrics derived from Oral Glucose Tolerance Tests (OGTTs) or Mixed Meal Tolerance Tests (MMTTs). Key parameters include:
- Homeostatic Model Assessment of Insulin Resistance (HOMA-IR): A simple index calculated from fasting glucose and insulin levels, providing an estimate of insulin resistance. The formula is $HOMA-IR = rac{Fasting Insulin (mIU/L) imes Fasting Glucose (mmol/L)}{22.5}$. Higher values indicate greater insulin resistance, which can influence the magnitude of postprandial insulin responses [3].
- Insulin Sensitivity Index (ISI): Derived from OGTTs (e.g., Matsuda index, Stumvoll index) or more complex intravenous tests, ISI quantifies how effectively insulin clears glucose from the bloodstream. A lower ISI signifies impaired insulin sensitivity [2, 3].
- Incremental Area Under the Curve (iAUC) for Glucose and Insulin: This metric quantifies the total excursion of glucose and insulin levels above fasting baseline over a specified postprandial period. Larger iAUC values for insulin, particularly after high-carbohydrate meals, are strongly associated with the proposed mechanism for somnolence [3].
- Glycemic Index (GI) and Glycemic Load (GL): These indices categorize foods based on their impact on blood glucose. High GI/GL foods lead to larger and faster glucose and insulin excursions, thus predicting a greater potential for somnolence [3].
Neurotransmitter Precursor Availability is often assessed indirectly:
- Tryptophan Availability Ratio: This ratio, calculated as Free Tryptophan / (Sum of competing LNAAs like tyrosine, phenylalanine, leucine, isoleucine, valine), is a critical predictor of brain tryptophan uptake. Postprandial insulin spikes can increase this ratio, enhancing the potential for serotonin synthesis [2, 3].
Hormonal Responses are quantified by measuring plasma levels of key hormones at various time points post-meal:
- Insulin, CCK, Ghrelin, and Leptin: Serial measurements of these hormones provide a detailed profile of the body's response to different meal compositions [2, 3].
Quantitative Models are employed to integrate these data:
- Glucose-Insulin Dynamics Models: Mathematical models, such as Bergman's Minimal Model, quantitatively describe the interaction between glucose and insulin, allowing for precise estimation of insulin sensitivity (S_I) and glucose effectiveness (S_G) [3]. These models provide a robust framework for understanding the metabolic consequences of different carbohydrate loads.
- Tryptophan Transport Models: These models aim to simulate the competition of amino acids for transport across the blood-brain barrier, incorporating factors like insulin levels to predict changes in brain tryptophan availability. They help to mechanistically link metabolic responses to potential neurochemical changes [3].
- Physiologically Based Pharmacokinetic (PBPK) Models: These advanced computational models simulate the absorption, distribution, metabolism, and excretion of nutrients and hormones within the body. They can be adapted to model complex nutrient-hormone-neurotransmitter interactions, allowing for in silico prediction of postprandial somnolence under various dietary scenarios [3].
Limitations and Research Gaps: Despite these advancements, significant limitations persist. Direct in vivo measurement of brain serotonin synthesis rates in humans postprandially remains challenging, relying heavily on indirect proxies [2]. The complex interplay of multiple hormones (insulin, CCK, ghrelin, leptin) and their combined effects on arousal are not fully elucidated [1, 2]. Furthermore, substantial inter-individual variability in responses to identical meals necessitates the development of personalized models that account for genetic predispositions, gut microbiome composition, and habitual dietary patterns [2]. Emerging research directions include leveraging advanced neuroimaging, omics approaches, and personalized nutrition strategies to gain a more comprehensive understanding of postprandial somnolence.
2. Dietary Factors and Modern Food Consumption's Impact
The pervasive experience of 'food coma,' or postprandial somnolence, is increasingly being linked to the fundamental shifts in global dietary patterns over recent decades. Modern food consumption is characterized by a high prevalence of processed items, refined carbohydrates, and large portion sizes, often consumed irrespective of natural physiological rhythms. This section critically examines how these contemporary dietary factors, particularly the composition and consumption habits related to modern food products, contribute to the exacerbation of post-meal drowsiness. We will delve into the specific mechanisms by which processed foods and high-glycemic meals influence metabolic responses, explore the interplay of meal size and timing within the context of modern lifestyles, and discuss the metrics used to quantify these dietary impacts, alongside the inherent challenges in establishing direct causal links.
2.1 Processed Foods and High-Glycemic Meals: Fueling Drowsiness
The modern food landscape is dominated by highly processed foods, which are often engineered for palatability, shelf-life, and convenience, frequently at the expense of nutritional integrity. These foods are typically rich in refined carbohydrates, unhealthy fats (such as saturated and trans fats), sodium, and a host of artificial additives. The manufacturing processes involved in creating processed foods often strip away natural fibers, essential micronutrients, and other beneficial compounds found in whole foods. This alteration in composition has profound implications for the body's metabolic response post-consumption.
Refined carbohydrates, such as those found in white bread, sugary cereals, pastries, and many processed snacks, are rapidly digested and absorbed into the bloodstream. This leads to sharp and rapid spikes in blood glucose levels. In response to this hyperglycemia, the pancreas releases a surge of insulin, a hormone crucial for facilitating glucose uptake by cells. However, this robust insulin response can overshoot, leading to a subsequent rapid drop in blood glucose levels, a phenomenon known as reactive hypoglycemia. This blood sugar crash is a primary driver of the fatigue, lethargy, and cognitive impairment associated with food coma. Furthermore, the insulin surge can promote the uptake of other amino acids into muscle tissue, relatively increasing the concentration of tryptophan in the bloodstream. Tryptophan is an essential amino acid that can cross the blood-brain barrier and serves as a precursor to serotonin and melatonin, neurotransmitters intimately involved in mood regulation, relaxation, and sleep induction [4]. Thus, meals high in refined carbohydrates can directly contribute to the physiological cascade leading to drowsiness.
The low fiber content characteristic of many processed foods exacerbates this effect. Fiber slows down gastric emptying and the rate of glucose absorption, acting as a buffer against rapid blood sugar fluctuations. Without adequate fiber, the digestive process is accelerated, leading to quicker absorption and a more pronounced glycemic response. This contributes to a faster onset and potentially more intense experience of post-meal sleepiness [4].
Beyond macronutrient composition, the role of food additives in inducing food coma, while less established, warrants consideration. Additives such as artificial sweeteners, flavor enhancers (like monosodium glutamate - MSG), emulsifiers, and preservatives are ubiquitous in processed foods. While robust scientific consensus on their direct causal role in promoting sleepiness is limited, some research and anecdotal reports suggest potential influences. For instance, certain artificial sweeteners have been linked to alterations in gut microbiota, which can, in turn, affect mood and energy levels. MSG has been anecdotally associated with fatigue in sensitive individuals, although scientific evidence for this specific effect in the context of food coma is scarce. The cumulative effect of consuming multiple additives in highly processed modern foods may contribute to a general sense of malaise or exacerbate existing susceptibilities to post-meal drowsiness in some individuals. However, it is crucial to emphasize that the primary drivers of food coma, based on current evidence, are the metabolic responses to refined carbohydrates and the overall meal composition rather than specific additives in isolation [4].
2.2 Meal Size, Timing, and Lifestyle Context: The Modern Predicament
Beyond the intrinsic properties of the food itself, the extrinsic factors of meal size and timing, deeply intertwined with modern lifestyle habits, significantly modulate the experience and prevalence of food coma. The sheer volume of food consumed and the temporal relationship of meals to natural biological rhythms play critical roles in dictating the intensity of postprandial somnolence.
Meal Size: Larger meals, irrespective of their specific composition, inherently demand a greater digestive effort from the body. The process of digestion is metabolically demanding, requiring significant energy expenditure and diverting blood flow from other bodily functions, including cognitive processes, towards the gastrointestinal tract. This diversion of blood flow can lead to a temporary reduction in alertness and a feeling of heaviness or lethargy. Furthermore, larger meals often trigger a more substantial release of satiety hormones, such as cholecystokinin (CCK) and peptide YY (PYY). These hormones, while crucial for signaling fullness, have also been implicated in promoting sleepiness and reducing overall arousal [5]. The modern trend towards larger portion sizes, prevalent in restaurant dining, convenience foods, and even home cooking, directly contributes to a more pronounced food coma experience. The physical distension of the stomach from a large meal can also contribute to discomfort and a general sense of sluggishness.
Meal Timing and Lifestyle Context: The timing of food consumption relative to an individual's natural circadian rhythms is another critical factor. Consuming large or heavy meals close to natural sleep periods, such as late in the evening, can synergize with the body's endogenous drive for sleep. As the day progresses, the body naturally prepares for rest, and a substantial digestive load at this time can amplify feelings of sleepiness. Modern lifestyles, characterized by irregular work schedules, late-night social activities, and the pervasive availability of food at all hours, often lead to meal patterns that are misaligned with these natural rhythms. For instance, late-night snacking or large evening meals are common, making individuals more susceptible to pronounced post-meal drowsiness during times when their alertness would naturally be declining [4].
The interplay between meal size, timing, and lifestyle is particularly evident in scenarios like large holiday meals. These events often involve consuming exceptionally large quantities of calorie-dense, carbohydrate-rich, and fat-laden foods, typically in the late afternoon or evening. This combination of excessive volume, specific food composition, and suboptimal timing creates a perfect storm for inducing severe postprandial somnolence, often leading to the characteristic 'food coma' that follows such feasts [5]. The modern context, therefore, not only influences what we eat but also when and how much, creating an environment where food coma is more likely to occur and be experienced intensely.
2.3 Assessment of Dietary Impact: Metrics, Challenges, and Variability
Quantifying the dietary contribution to postprandial somnolence involves employing various metrics that assess the physiological impact of food consumption. However, establishing direct, universally applicable causal links between diet and food coma is complicated by methodological challenges and significant individual variability.
Metrics for Assessing Dietary Impact: Several metrics are used to evaluate how different foods and meals might induce sleepiness:
- Glycemic Index (GI): The GI ranks carbohydrate-containing foods based on their potential to raise blood glucose levels after consumption. Foods with a high GI (e.g., >70) cause a rapid and significant rise in blood sugar, leading to a robust insulin response. This is a key factor in the tryptophan-serotonin-melatonin pathway associated with food coma [5].
- Glycemic Load (GL): GL takes into account both the GI of a food and the amount of carbohydrate in a typical serving. It provides a more practical measure of a food's overall impact on blood glucose. A high GL meal is more likely to trigger significant metabolic shifts contributing to drowsiness [5].
- Insulinemic Index (II): This metric measures the insulin response elicited by a food, independent of its carbohydrate content. Some foods, particularly those high in protein or fat, can stimulate a substantial insulin release even if their GI is moderate. Since insulin is central to facilitating tryptophan uptake into the brain, the II offers a complementary perspective to GI/GL in assessing sleep-inducing potential [5].
- Macronutrient Ratios: While not a single metric, the proportion of carbohydrates (especially refined), fats, and proteins in a meal is critical. Meals high in both refined carbohydrates and fats are consistently linked to increased postprandial somnolence due to the combined effects on glucose regulation, insulin response, and digestive load [5].
Methodological Challenges: Despite these metrics, research into food coma faces several hurdles:
- Defining and Quantifying 'Food Coma': There is a lack of standardized, objective measures for postprandial somnolence. Most assessments rely on subjective self-reports of sleepiness, which can be influenced by numerous external factors such as stress, sleep deprivation, mood, and expectations. Objective measures like polysomnography or continuous EEG are rarely employed in dietary studies due to practical limitations [6].
- Controlling for Confounding Factors: Isolating the specific impact of diet from other lifestyle variables is extremely challenging. Factors such as sleep quality, physical activity levels, stress, hydration, and the use of certain medications can all influence post-meal energy levels and perceived sleepiness [6]. Real-world observational studies often struggle to adequately control for these confounds.
- Dietary Complexity and Additives: Modern diets are incredibly complex, making it difficult to pinpoint the exact contribution of individual food components versus the overall dietary pattern. The role of specific food additives, while anecdotally linked to fatigue, lacks robust empirical evidence and requires more targeted research to establish direct causal pathways [4][6].
- Blinding in Intervention Studies: It is often impossible to blind participants to the type of food they are consuming, particularly when comparing vastly different meal compositions (e.g., whole foods versus highly processed meals). This lack of blinding can introduce placebo effects or biases in subjective reporting.
Individual Variability: A significant limitation in generalizing findings is the substantial individual variability in metabolic responses to food. Genetic predispositions, the composition of an individual's gut microbiome, their level of insulin sensitivity, hormonal profiles, and even long-term dietary adaptation can all influence how a person responds to a given meal. For example, an individual with high insulin sensitivity might experience a less pronounced blood sugar crash after a high-GI meal compared to someone with insulin resistance [4][6]. This inherent variability means that a meal that induces significant drowsiness in one person may have little effect on another, complicating the development of universal predictive models for food coma potential.
3. Health Implications and Strategies for Mitigation
Post-meal drowsiness, commonly referred to as 'food coma' or postprandial somnolence, can range from a mild, transient feeling of lethargy to a debilitating experience that significantly impacts daily functioning. While often attributed solely to the composition of a meal, pronounced or persistent postprandial somnolence can be indicative of underlying health conditions that warrant attention. Understanding these associations is crucial for effective management and for distinguishing typical physiological responses from symptoms of more serious medical issues. This section delves into the health conditions that can exacerbate food coma, explores evidence-based strategies for mitigation, and outlines when professional medical advice should be sought.
3.1 Associated Health Conditions
Several physiological and pathological states can amplify or mimic the effects of food coma, making post-meal drowsiness more severe or persistent. Recognizing these links is the first step toward targeted intervention.
Insulin Resistance and Reactive Hypoglycemia: A primary physiological driver behind exacerbated food coma is the interplay between insulin resistance (IR) and reactive hypoglycemia. In individuals with IR, the body's cells do not respond efficiently to insulin, leading to elevated blood glucose levels after a meal (postprandial hyperglycemia). The pancreas compensates by releasing an excessive amount of insulin to try and lower blood sugar. This compensatory hyperinsulinemia can then cause blood glucose levels to drop too rapidly and too low, resulting in reactive hypoglycemia, typically defined as a blood glucose level below 70 mg/dL occurring 2-4 hours after eating [8, 9]. This rapid decline in blood glucose deprives the brain of its primary fuel source, leading to symptoms of fatigue, dizziness, confusion, and pronounced drowsiness [8]. Furthermore, high insulin levels can facilitate the transport of tryptophan into the brain, increasing the synthesis of serotonin and melatonin, neurotransmitters associated with sleepiness [8]. Quantitative measures like the Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) suggest a positive correlation between higher scores (indicating greater IR) and the severity of postprandial fatigue. Additionally, the amplitude of glucose excursions, as measured by continuous glucose monitoring (CGM), where rapid spikes (>40 mg/dL rise) are followed by sharp declines, is a stronger predictor of postprandial somnolence than fasting glucose alone [8]. Chronic insulin resistance is a precursor to metabolic syndrome and type 2 diabetes, making persistent post-meal drowsiness a potential early warning sign [7].
Sleep Disorders: Pre-existing sleep disorders significantly exacerbate postprandial somnolence. Individuals with undiagnosed or poorly managed sleep apnea experience fragmented sleep and reduced oxygen saturation, leading to chronic daytime sleepiness that is often intensified after meals. The digestive process requires increased blood flow to the gastrointestinal tract, which can further strain an already compromised respiratory system during sleep episodes, leading to more pronounced fatigue the following day [7]. Similarly, narcolepsy, a neurological disorder characterized by excessive daytime sleepiness and sudden sleep attacks, can be mistaken for or worsened by typical food coma. The natural circadian dip in alertness that occurs in the early afternoon can trigger a sleep attack in individuals with narcolepsy, making the post-meal period particularly challenging [7]. Other circadian rhythm disorders, whether due to shift work, irregular sleep schedules, or other factors, can disrupt the body's internal clock, making individuals more sensitive to the natural decline in alertness that follows a meal [7].
Gastrointestinal Disorders: Certain gastrointestinal conditions can also contribute to post-meal fatigue. Conditions like Gastroesophageal Reflux Disease (GERD) or Irritable Bowel Syndrome (IBS) can cause discomfort, bloating, and impaired digestion, which can indirectly lead to lethargy. The body expends significant energy on digestion, and when this process is complicated by underlying GI issues, the resulting fatigue can be amplified. Furthermore, malabsorption issues or delayed gastric emptying associated with some GI disorders can prolong the digestive effort, contributing to a feeling of heaviness and sleepiness [7].
3.2 Dietary and Lifestyle Mitigation Strategies
Fortunately, many strategies can help reduce or eliminate post-meal drowsiness, focusing on dietary adjustments and lifestyle modifications. These approaches aim to stabilize blood glucose levels, improve metabolic function, and enhance overall energy regulation.
Dietary Adjustments: The cornerstone of mitigating food coma lies in optimizing meal composition and consumption habits.
- Balanced Macronutrient Distribution: Prioritizing meals that contain a balanced ratio of complex carbohydrates, lean protein, and healthy fats is crucial. Complex carbohydrates (e.g., whole grains, legumes, vegetables) are digested more slowly, leading to a gradual rise in blood glucose, unlike refined carbohydrates (e.g., white bread, sugary snacks) which cause rapid spikes and subsequent crashes [7]. Protein and healthy fats slow gastric emptying and enhance satiety, further contributing to stable energy levels. For example, pairing a lean protein source like grilled chicken or fish with a serving of quinoa and a side of steamed vegetables provides sustained energy. Limiting processed foods and sugary beverages is essential, as they are major contributors to blood sugar fluctuations [7].
- Portion Control: Consuming excessively large meals places a significant burden on the digestive system, diverting blood flow and metabolic resources away from other bodily functions, which can induce drowsiness. Opting for smaller, more frequent meals throughout the day can help manage the digestive load and maintain more consistent energy levels. Instead of three large meals, consider five to six smaller ones [7].
- Adequate Fiber Intake: Dietary fiber, particularly soluble fiber found in oats, beans, apples, and citrus fruits, plays a vital role in slowing down glucose absorption. This leads to a more gradual increase in blood sugar after eating, preventing the sharp peaks and subsequent energy crashes associated with high-glycemic meals [7]. Aiming for at least 25-30 grams of fiber per day is recommended.
- Hydration: Dehydration can significantly contribute to feelings of fatigue and lethargy. Ensuring adequate fluid intake, primarily water, throughout the day, and especially around mealtimes, can help maintain optimal physiological function and energy levels [7].
- Mindful Eating: Practicing mindful eating—paying attention to hunger and fullness cues, eating slowly, and savoring each bite—can improve digestion and prevent overeating, a common trigger for post-meal drowsiness. Chewing food thoroughly also aids digestion [7].
Lifestyle Modifications: Beyond diet, several lifestyle adjustments can profoundly impact post-meal alertness.
- Regular Physical Activity: Engaging in regular physical activity is paramount for improving insulin sensitivity and overall metabolic health. A brisk 15-20 minute walk after meals can be particularly beneficial. Exercise helps muscles utilize glucose for energy, reducing the amount of glucose circulating in the bloodstream and mitigating the need for large insulin responses. This post-meal activity aids in glucose uptake, promoting alertness and preventing the energy dip [7].
- Consistent Sleep Hygiene: Maintaining a regular sleep schedule, even on weekends, is critical for regulating the body's circadian rhythm and ensuring adequate restorative sleep. Aiming for 7-9 hours of quality sleep per night helps to reduce baseline daytime sleepiness, making individuals less susceptible to pronounced post-meal drowsiness [7]. Establishing a relaxing bedtime routine and ensuring a dark, quiet sleep environment are key components of good sleep hygiene.
- Stress Management: Chronic stress can disrupt hormonal balance, particularly cortisol levels, and negatively impact sleep quality, both of which can contribute to fatigue. Implementing stress-reduction techniques such as meditation, deep breathing exercises, yoga, or engaging in enjoyable hobbies can improve overall well-being and energy regulation [7].
- Limiting Alcohol and Sedatives: Alcohol and certain medications can induce drowsiness, and their effects can be amplified when consumed with meals. Moderating alcohol intake and being aware of the potential sedative effects of medications are important considerations [7].
| Strategy Category | Specific Strategy | Key Actionable Advice |
|---|---|---|
| Dietary | Balanced Macronutrient Intake | Combine complex carbohydrates, lean protein, and healthy fats in each meal. Limit refined sugars and processed foods. |
| Portion Control | Eat smaller, more frequent meals; avoid overeating. | |
| Fiber Intake | Increase consumption of fruits, vegetables, legumes, and whole grains. | |
| Hydration | Drink adequate water throughout the day, especially around meals. | |
| Mindful Eating | Eat slowly, chew thoroughly, and pay attention to hunger/fullness cues. | |
| Lifestyle | Regular Physical Activity | Engage in a 15-20 minute brisk walk after meals; aim for consistent daily exercise. |
| Consistent Sleep Schedule | Go to bed and wake up around the same time daily; ensure 7-9 hours of quality sleep. | |
| Stress Management | Practice relaxation techniques like meditation, deep breathing, or yoga. | |
| Limit Alcohol and Sedatives | Consume alcohol in moderation and be aware of medication side effects. |
3.3 When to Seek Professional Advice
While occasional post-meal drowsiness is normal, persistent, severe, or debilitating fatigue after eating warrants medical evaluation. It is essential to differentiate typical food coma from symptoms indicative of underlying health issues that require professional diagnosis and management.
Persistent and Severe Drowsiness: If post-meal drowsiness is so profound that it significantly interferes with daily activities, work, or social life, it is a clear signal to consult a healthcare provider. This level of fatigue may suggest an underlying condition that is not being adequately managed [7].
Associated Symptoms: The presence of other symptoms alongside post-meal drowsiness can point towards specific medical concerns:
- Symptoms suggestive of diabetes or pre-diabetes: Excessive thirst (polydipsia), frequent urination (polyuria), unexplained weight loss, increased hunger (polyphagia), and blurred vision can indicate dysregulated blood sugar control [7].
- Symptoms suggestive of sleep apnea: Loud snoring, observed pauses in breathing during sleep, gasping for air during sleep, morning headaches, and extreme daytime sleepiness (beyond post-meal fatigue) are critical indicators for sleep disorder evaluation [7].
- Symptoms suggestive of other metabolic or endocrine disorders: Unexplained weight changes, fatigue not solely related to meals, changes in mood or energy levels, or digestive disturbances that are persistent and severe may warrant investigation into thyroid function or other hormonal imbalances.
- Neurological symptoms: Dizziness, confusion, or cognitive impairment that occurs specifically after meals, especially if accompanied by other neurological signs, should be investigated.
Lack of Improvement with Lifestyle Changes: If individuals diligently implement the dietary and lifestyle modifications outlined above, yet their post-meal drowsiness persists or worsens, it strongly suggests that a more significant underlying issue may be present. A healthcare professional can conduct appropriate diagnostic tests, such as an oral glucose tolerance test (OGTT) to assess for reactive hypoglycemia or diabetes, or a polysomnography (sleep study) to diagnose sleep apnea, among other investigations [7, 9].
Seeking professional medical advice is a proactive step towards understanding the root cause of persistent food coma and ensuring overall health and well-being. Early diagnosis and management of associated health conditions can prevent long-term complications and significantly improve quality of life.
Conclusion and Future Directions
This research has systematically investigated the phenomenon of postprandial somnolence, commonly referred to as 'food coma,' examining its underlying physiological mechanisms, the impact of modern dietary habits, and strategies for mitigation. Our findings underscore that food coma is a complex physiological response, not merely a subjective feeling, stemming from the intricate interplay of macronutrient composition, hormonal signaling, and neural pathways. High-carbohydrate meals, particularly those with refined sugars, induce significant glucose and insulin spikes. This insulin surge indirectly enhances the brain's uptake of tryptophan, a precursor to serotonin and melatonin, thereby promoting relaxation and drowsiness. While fatty meals contribute to satiety and may influence sleep-wake cycles via hormones like cholecystokinin (CCK), their direct role in immediate somnolence is less pronounced than that of carbohydrates. Modern dietary patterns, characterized by the prevalence of processed foods and high-glycemic index meals, exacerbate these effects by creating frequent and exaggerated metabolic fluctuations. These diets often lack fiber and are rich in refined sugars and unhealthy fats, leading to amplified insulin responses and subsequent somnolence. Furthermore, meal size and timing, particularly when large meals are consumed close to natural sleep periods, can synergize with circadian rhythms to intensify post-meal lethargy. The research also highlighted that while food coma is a common experience, it can be exacerbated by underlying health conditions such as insulin resistance, where reactive hypoglycemia can significantly contribute to fatigue. Practical mitigation strategies, including dietary adjustments (balanced macronutrients, increased fiber, portion control), and lifestyle modifications (regular physical activity, consistent sleep hygiene), offer effective avenues for individuals to manage and reduce postprandial somnolence. Seeking professional medical advice is crucial when symptoms are severe, persistent, or accompanied by other concerning health indicators.
The primary theoretical contribution of this research lies in synthesizing the current understanding of the neuro-metabolic pathways driving postprandial somnolence, emphasizing the critical role of the tryptophan-serotonin-melatonin axis modulated by insulin response. Methodologically, the review highlights the utility of quantitative metrics such as Glycemic Index, Glycemic Load, and Insulinemic Index in assessing dietary impacts, while also pointing to the need for more objective measures of somnolence. The practical value is evident in the actionable strategies provided for individuals and potentially for public health recommendations concerning dietary guidelines. The research offers a holistic perspective, moving beyond a simplistic view of 'food coma' to a scientifically grounded understanding of its determinants and management.
Despite the comprehensive review, several limitations persist. The significant individual variability in response to dietary intake, influenced by genetics, gut microbiome composition, and hormonal profiles, remains a substantial challenge in establishing universal predictive models for food coma severity. Direct quantification of brain neurotransmitter flux in humans in response to specific meals is methodologically complex and often relies on indirect proxies. Furthermore, the precise contribution of individual food additives, beyond their presence in highly processed foods, requires more robust empirical investigation. The subjective nature of reporting somnolence, while common, introduces variability and necessitates further development of objective assessment tools for acute postprandial states.
Future research should prioritize investigating personalized nutrition approaches, tailoring dietary recommendations based on an individual's metabolic profile, genetic predispositions, and gut microbiome. Further exploration of the gut-brain axis, including the role of gut peptides and microbial metabolites in modulating sleep-wake cycles, is essential. Developing more objective and standardized measures for assessing postprandial somnolence would significantly advance the field. Additionally, longitudinal studies examining the long-term health implications of chronic postprandial somnolence, particularly in relation to metabolic disorders and cognitive function, are warranted. The integration of chrononutrition principles, focusing on the interaction between meal timing and circadian rhythms, also presents a promising avenue for mitigating food coma and optimizing overall well-being. Ultimately, a deeper understanding of these interconnected factors will pave the way for more effective, individualized interventions.
References
[1] llm_self_research
- Query: Physiological mechanisms of postprandial somnolence, food coma, macronutrients, insulin, serotonin, melatonin, neural pathways, digestive system, sleep regulation
- Summary: Postprandial somnolence, commonly known as 'food coma,' is a temporary state of drowsiness and reduced alertness experienced after eating. This phenomenon is influenced by the interplay of macronutrients, hormonal responses, and neural signaling.
High-carbohydrate meals, especially those with refin...
[2] llm_self_research
- Query: Investigate quantitative models or specific physiological parameters used to measure the impact of macronutrient intake on postprandial somnolence, including the role of insulin sensitivity, glucose metabolism kinetics, and neurotransmitter synthesis rates. Explore research methodologies and experimental designs used to study food coma, and identify current gaps in understanding and emerging research directions.
- Summary: Postprandial somnolence, or 'food coma,' is a complex physiological response to food intake influenced by macronutrient composition, hormonal signaling, and neural pathways. Quantitative models and physiological parameters are crucial for understanding this phenomenon. Key parameters include Insulin...
[3] llm_self_research
- Query: Detailed physiological parameters and quantitative models used to measure and understand postprandial somnolence, including specific metrics for insulin sensitivity, glucose metabolism kinetics, and hormonal responses. Also, explore practical scenarios where these models are applied to assess or manage food coma.
- Summary: Postprandial somnolence, colloquially known as 'food coma,' is a multifaceted physiological phenomenon driven by the integration of macronutrient intake, hormonal fluctuations, and neural signaling. The primary metabolic driver is the postprandial glucose-insulin cascade. High-carbohydrate intake in...
[4] llm_self_research
- Query: Dietary factors and modern food consumption impact on food coma, processed foods, high glycemic index meals, food additives, meal timing, meal size, post-meal sleepiness
- Summary: The phenomenon of 'food coma,' or postprandial somnolence, is significantly influenced by modern dietary factors and consumption patterns. Processed foods, characterized by refined carbohydrates, unhealthy fats, and low fiber, contribute to rapid blood glucose spikes and subsequent crashes, potentia...
[5] llm_self_research
- Query: Explore specific examples and case studies of modern food consumption patterns (e.g., typical fast food meals, processed snack consumption, large holiday meals) and their documented impact on postprandial somnolence. Investigate if there are any quantitative models or metrics used to assess the 'food coma' potential of specific meals or dietary patterns.
- Summary: Postprandial somnolence, colloquially known as 'food coma,' is a complex physiological response driven by modern dietary patterns and metabolic processes. The phenomenon is primarily linked to the consumption of high-glycemic index (GI) and high-glycemic load (GL) foods, which trigger rapid glucose ...
[6] llm_self_research
- Query: Explore real-world examples and case studies illustrating how specific modern dietary patterns (e.g., frequent consumption of fast food, processed snacks, large holiday meals) lead to pronounced food coma symptoms. Additionally, investigate the limitations of current research on food coma, including methodological challenges, individual variability, and emerging areas of study.
- Summary: Modern dietary patterns, particularly the frequent consumption of fast food, processed snacks, and large holiday meals, significantly contribute to pronounced 'food coma' symptoms, scientifically known as postprandial somnolence. These dietary habits lead to pronounced physiological responses primar...
[7] llm_self_research
- Query: Underlying health conditions associated with excessive food coma, such as insulin resistance or sleep disorders, and evidence-based strategies for mitigating post-meal drowsiness including dietary adjustments and lifestyle modifications.
- Summary: Post-meal drowsiness, commonly known as 'food coma' or postprandial somnolence, is a physiological response influenced by metabolic, circadian, and gastrointestinal factors. Underlying health conditions often exacerbate this state, including insulin resistance, where impaired glucose metabolism lead...
[8] llm_self_research
- Query: Explore quantitative measures and specific metabolic pathways linking insulin resistance, reactive hypoglycemia, and postprandial somnolence. Detail the complex interactions between gut microbiome composition, genetic predispositions, and the severity of food coma symptoms, citing research that quantifies these relationships.
- Summary: Metabolic Pathways and Quantitative Links
Insulin Resistance (IR) and Postprandial Somnolence (PS):
Mechanism: IR impairs the translocation of GLUT4 transporters to the cell membrane, leading to prolonged postprandial hyperglycemia. The subsequent compensatory hyperinsulinemia triggers a rapid gluco...
[9] llm_self_research
- Query: Quantitative thresholds for postprandial glucose variability and insulin response in relation to somnolence; clinical limitations and long-term efficacy of lifestyle interventions for postprandial somnolence; emerging research on gut-brain axis signaling (e.g., GLP-1, PYY) in post-meal fatigue.
- Summary: Quantitative Thresholds for Postprandial Glucose Variability and Insulin Response in Relation to Somnolence
While precise, universally agreed-upon quantitative thresholds for postprandial glucose variability and insulin response directly linked to somnolence are not definitively established in gene...