Neurobiological and Systemic Etiologies of Depression: A Physical and Environmental Analysis
Introduction
The pervasive nature of depression, affecting millions globally, necessitates a deeper understanding beyond its well-documented psychological manifestations. Recent trends in medical research increasingly point towards the intricate interplay between mental health and the physical body, suggesting that depression may manifest with significant, measurable somatic and neurobiological underpinnings. This growing recognition highlights a critical gap in current understanding, particularly concerning the direct physical sequelae of depressive states, their impact on organ systems, and the potential for organ-specific dysfunctions to contribute to or arise from depression. Addressing this complexity is urgent, as a comprehensive view is essential for developing more effective diagnostic and therapeutic strategies that acknowledge the holistic nature of human health.
This research report specifically addresses the challenge of delineating the physical, non-psychological dimensions of depression. Existing research often emphasizes the psychological and emotional aspects, potentially overlooking or underestimating the direct physical alterations that occur within the brain and body. This report aims to bridge this gap by investigating the neurobiological structural changes associated with depression, exploring how these changes might affect specific bodily organs, and examining whether dysfunction in certain organs could be a causal factor in the development of depressive symptoms. The core objectives are to identify and analyze the physical correlates of depression, understand its impact on organ systems, and explore the potential for organ-related etiologies.
The scope of this research is structured around three key dimensions: the neurobiological correlates of depression and their potential impact on organ systems; the influence of environmental and nutritional factors on the pathophysiology of depressive states; and the examination of organ-related etiologies as potential causes or significant contributors to depression. These dimensions are logically interconnected, forming a comprehensive framework to explore the physical underpinnings of depression. The neurobiological focus will lay the groundwork by examining brain changes, which will then inform the investigation into how these changes might affect organ function. Concurrently, the role of external factors like diet and environment will be analyzed for their impact on these neurobiological and physiological pathways. Finally, the report will critically assess the reciprocal relationship between organ health and depression, considering whether conditions in organs like the thyroid or gut can directly precipitate depressive symptoms.
To facilitate a clear and structured understanding of these complex issues, this report is organized into distinct sections. Following this introduction, the subsequent sections will delve into the detailed findings related to neurobiological correlates and organ impact, environmental and nutritional influences, and organ-related etiologies. Each section is designed to build upon the previous one, offering a progressive exploration of the physical dimensions of depression. This organization is intended to guide the reader through the multifaceted aspects of the research, providing a comprehensive overview of the current understanding and highlighting areas for future investigation.
1. Neurobiological Correlates of Depression
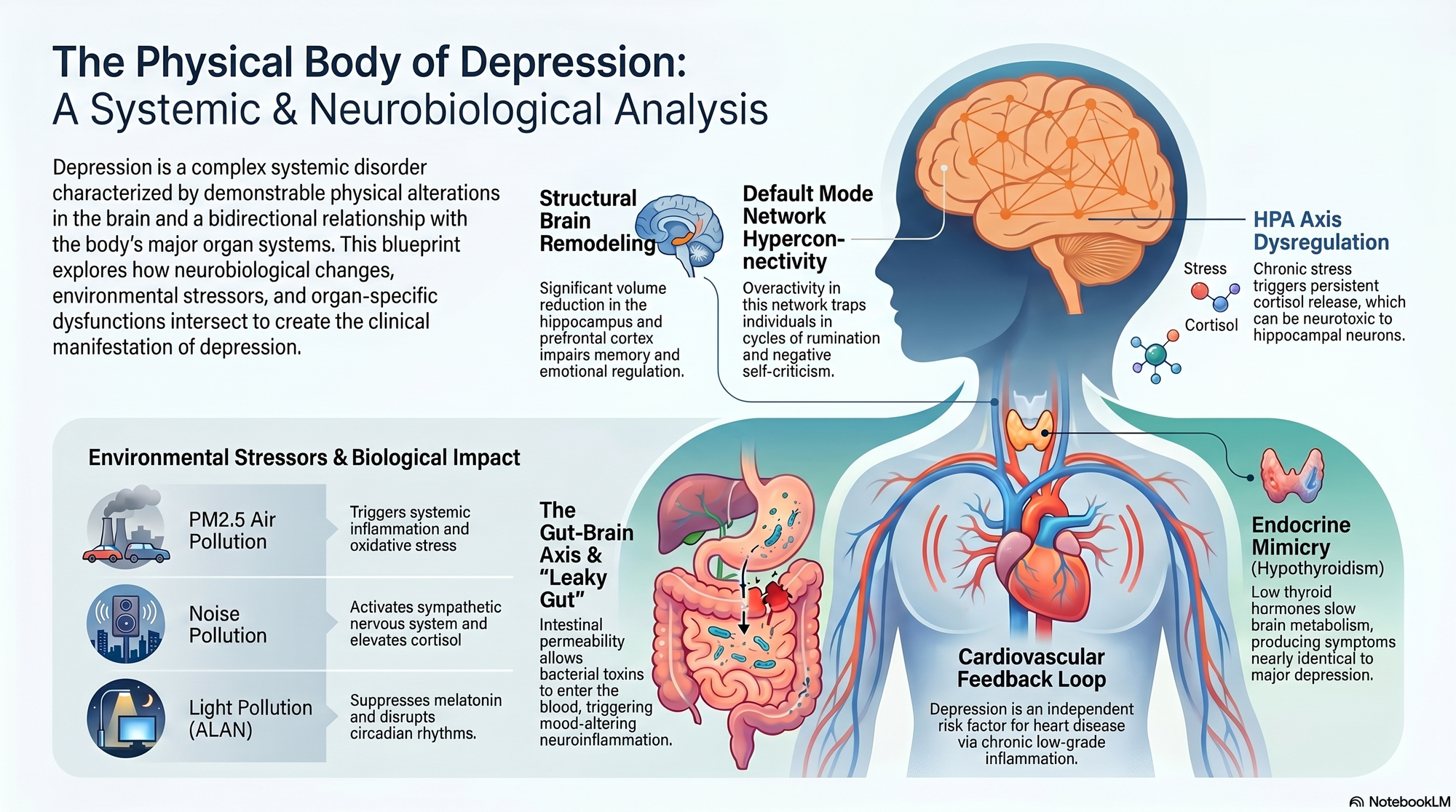
Depression, beyond its subjective experience, is increasingly understood as a complex neurobiological disorder characterized by demonstrable physical alterations within the brain. This section shifts the focus from psychological interpretations to the objective, physiological markers that underpin the condition. By examining structural modifications, functional neurochemistry, network dynamics, and the methodologies employed to assess these changes, we can gain a deeper insight into the physical reality of depression and its pervasive impact.
1.1 Structural Brain Modifications
Neuroimaging studies have consistently revealed structural alterations in key brain regions implicated in mood regulation, emotional processing, and cognitive control in individuals diagnosed with depression. These modifications, often quantifiable through comparative analyses of magnetic resonance imaging (MRI) data, highlight a departure from normative brain anatomy and integrity.
Hippocampus: A significant body of research points to a reduction in hippocampal volume in individuals with depression. This region, critical for learning, memory formation, and the regulation of the stress response, is particularly vulnerable. Studies using volumetric analysis of MRI scans have reported decreased gray matter volume, especially in subfields like the CA3 and dentate gyrus. This reduction is often associated with impaired neurogenesis (the birth of new neurons) and synaptic plasticity, the ability of neural connections to strengthen or weaken over time, which are vital for adaptive responses and mood stability [1]. The hippocampus's role in the hypothalamic-pituitary-adrenal (HPA) axis feedback loop means its dysfunction can perpetuate a cycle of stress hormone dysregulation, further exacerbating depressive symptoms. However, it is crucial to acknowledge the heterogeneity in findings, with some studies reporting no significant hippocampal volume reduction, suggesting that these changes may not be universal across all individuals with depression or that subtle alterations might be missed by current imaging techniques [1].
Amygdala: In contrast to the hippocampus, the amygdala, a key center for processing emotions, particularly fear and threat, often exhibits structural changes that correlate with depressive states. Neuroimaging studies, including fMRI, frequently report increased amygdala volume and heightened activity, especially in response to negative emotional stimuli. This hyperactivity is thought to contribute to the characteristic hypervigilance, anxiety, and exaggerated negative emotional reactivity seen in many individuals with depression. The amygdala's intricate connections with the HPA axis mean its dysregulation can also drive stress responses. Nevertheless, some research indicates amygdala hypoactivity in specific depressive subtypes or suggests that observed changes are more reflective of deficits in emotional regulation rather than absolute size increases, underscoring the complexity of its role [1].
Prefrontal Cortex (PFC): The PFC, responsible for executive functions such as planning, decision-making, working memory, and emotional regulation, shows notable structural alterations in depression. Reductions in gray matter volume and altered functional connectivity have been observed in subregions like the dorsolateral PFC (dlPFC), ventromedial PFC (vmPFC), and orbitofrontal cortex (OFC). These changes are linked to impaired cognitive control, difficulties in initiating and sustaining goal-directed behavior, flattened affect, and altered reward processing, particularly in the vmPFC and OFC [1]. The dlPFC's role in cognitive reappraisal and inhibitory control means its dysfunction can contribute to rumination and an inability to disengage from negative thoughts. While many studies report reduced gray matter, some task-based fMRI studies have shown increased activity in specific PFC regions during certain cognitive tasks, highlighting the complex, context-dependent nature of PFC involvement in depression [1].
Anterior Cingulate Cortex (ACC): The ACC, a region involved in error detection, conflict monitoring, and the integration of cognitive and emotional information, also exhibits structural changes. Reduced gray matter volume and altered activity, particularly in the dorsal ACC (dACC), are frequently reported. The dACC's role in cognitive control suggests its dysfunction may contribute to difficulties in modulating emotional responses and an inability to shift attention away from negative stimuli, fostering rumination. Conversely, some research indicates ACC hyperactivity in response to negative stimuli, suggesting its involvement in the processing of negative affect and distress [1]. The distinction between dorsal and ventral ACC subregions and their specific roles further complicates the interpretation of these findings [1].
Collectively, these structural modifications in the hippocampus, amygdala, PFC, and ACC paint a picture of a brain whose circuitry for emotion regulation, cognitive control, and stress response is physically altered, providing a neurobiological basis for the multifaceted symptoms of depression.
1.2 Functional Neurochemistry and Network Dynamics
Beyond structural changes, depression is profoundly characterized by functional disruptions in neurochemistry and the dynamic interplay of neural networks. These functional alterations are critical in understanding the pervasive mood, cognitive, and somatic symptoms associated with the disorder.
Neurotransmitter Dysregulation: A cornerstone of biological theories of depression involves the dysregulation of key neurotransmitter systems. Imbalances in monoamines, including serotonin (5-HT), norepinephrine (NE), and dopamine (DA), are central. Reduced levels or impaired signaling of serotonin and norepinephrine are associated with mood disturbances, anxiety, and sleep problems. Dopamine dysregulation, particularly in the mesolimbic reward pathways, is linked to anhedonia (loss of pleasure) and reduced motivation. Furthermore, the glutamatergic system, the brain's primary excitatory neurotransmitter system, and the inhibitory gamma-aminobutyric acid (GABA) system are also implicated. Dysregulation of glutamate can lead to excitotoxicity and impaired synaptic plasticity, while GABAergic deficits can contribute to anxiety and heightened neural excitability. While early research focused on absolute levels, current understanding emphasizes receptor sensitivity, transporter function, and the complex interplay between these systems. Methodological challenges, such as the difficulty of directly measuring neurotransmitter levels in vivo in humans and the confounding effects of medication, contribute to ongoing debates regarding the precise nature and directionality of these imbalances [1].
Hypothalamic-Pituitary-Adrenal (HPA) Axis Dysfunction: The HPA axis is the body's central stress response system. In depression, this system is often chronically activated and dysregulated, leading to elevated levels of cortisol, the primary stress hormone. This hypercortisolemia can be detected through blood or salivary tests. The HPA axis dysfunction contributes to impaired negative feedback mechanisms, meaning the system fails to adequately shut down after a stressor. Chronic exposure to elevated cortisol has detrimental effects, including neurotoxicity in the hippocampus (as discussed above), immune suppression, metabolic disturbances, and cardiovascular problems. This persistent activation of the stress response system creates a state of allostatic load, where the body's adaptive systems are overwhelmed, contributing to both psychological and physical symptoms of depression [1]. Some individuals with depression, however, may exhibit a blunted HPA axis response, indicating a more complex pattern of dysregulation than simple hypercortisolemia [1].
Altered Default Mode Network (DMN) Activity: The DMN is a network of brain regions (including the medial prefrontal cortex, posterior cingulate cortex, and angular gyrus) that is most active when an individual is not focused on the outside world, such as during mind-wandering, self-referential thought, and rumination. In depression, studies using resting-state fMRI consistently show increased connectivity and activity within the DMN. This hyperconnectivity is strongly associated with the characteristic rumination, excessive self-criticism, and preoccupation with past negative experiences that are hallmarks of depressive episodes. The DMN's overactivity can trap individuals in cycles of negative thinking, making it difficult to disengage from internal distress and engage with external reality or problem-solving. The precise nature of DMN alterations can vary, with some circuits showing increased connectivity while others might show decreased connectivity depending on the specific depressive subtype and task [1].
Impaired Emotion Regulation Circuits: Depression is characterized by a profound deficit in the ability to regulate emotions. This is reflected in the dysfunctional interplay between limbic regions, such as the amygdala (involved in emotion generation), and prefrontal control regions, like the dlPFC and ACC (involved in modulating emotional responses). Neuroimaging studies reveal that in depression, the prefrontal cortex often fails to adequately inhibit or regulate the activity of the amygdala, leading to heightened emotional reactivity and difficulty in down-regulating negative affect. This imbalance between bottom-up emotional signaling (from limbic areas) and top-down cognitive control (from the PFC) is a critical functional deficit contributing to the emotional dysregulation observed in depression [1].
These functional neurochemical and network dynamics underscore that depression is not merely a matter of mood but a profound disruption of brain systems responsible for emotion processing, stress response, cognitive control, and self-referential thought.
1.3 Methodologies in Neurobiological Assessment
The investigation into the neurobiological correlates of depression relies on a sophisticated array of neuroimaging techniques, neurophysiological assessments, and biochemical analyses. These methodologies allow researchers to quantify structural and functional brain changes, providing objective data to complement subjective clinical reports. However, translating these findings into robust clinical diagnostic criteria and predictive models remains an ongoing challenge.
Functional Magnetic Resonance Imaging (fMRI): fMRI is a cornerstone for assessing brain function by detecting changes in blood oxygenation (the BOLD signal), which correlates with neural activity. In depression research, fMRI is employed in both task-based paradigms and resting-state scans. Task-based fMRI examines brain activity during specific cognitive or emotional challenges (e.g., viewing emotional faces, performing executive function tasks), allowing researchers to identify hypo- or hyperactive regions and altered task-related connectivity. Resting-state fMRI, conversely, measures spontaneous brain activity and functional connectivity when a participant is at rest, revealing intrinsic network dynamics, such as the overactivity of the DMN [1, 3]. Analysis typically involves extensive preprocessing (motion correction, spatial smoothing, normalization) followed by statistical modeling, most commonly the General Linear Model (GLM), to identify significant group differences in activation or connectivity. Advanced techniques like Independent Component Analysis (ICA) are used to identify distinct functional networks, while seed-based correlation analysis quantifies connectivity between specific regions [2, 3].
Electroencephalography (EEG): EEG measures the electrical activity of the brain via electrodes placed on the scalp. Its primary advantage is its exceptional temporal resolution, allowing for the study of rapid neural processes. In depression, EEG is used to examine alterations in brain oscillations (e.g., alpha, theta, gamma bands) and Event-Related Potentials (ERPs). Time-frequency analysis, employing methods like the Short-Time Fourier Transform (STFT) or wavelet transforms, reveals changes in the power and coherence of different frequency bands, such as altered alpha asymmetry in frontal regions, which has been linked to mood states [1, 3]. ERPs, which are averaged EEG responses time-locked to specific stimuli, can reveal deficits in cognitive processing, such as attentional biases or impaired error monitoring, by examining the amplitude and latency of components like the P300 or N400 [2, 3]. Source localization algorithms, such as LORETA, attempt to estimate the underlying brain regions generating the scalp-recorded electrical activity [3].
Diffusion Tensor Imaging (DTI): DTI is a specialized MRI technique used to assess the integrity and organization of white matter tracts, the brain's communication pathways. By measuring the diffusion of water molecules, DTI can infer the microstructural properties of white matter. Key metrics include Fractional Anisotropy (FA), which reflects the directionality of water diffusion (higher FA generally indicates more organized and intact white matter), and Mean Diffusivity (MD), which measures the overall magnitude of water diffusion (higher MD can indicate white matter damage or disorganization) [1, 2]. In depression, DTI studies have reported alterations in FA and MD in various white matter tracts, suggesting disruptions in structural connectivity between brain regions involved in mood regulation and cognitive control. Tractography algorithms are used to reconstruct these pathways, allowing for the quantitative assessment of connectivity strength and integrity [2].
Biochemical and Hormonal Assays: Beyond neuroimaging, biochemical markers are crucial. Cerebrospinal fluid (CSF) analysis, PET imaging with specific radioligands, and blood/saliva tests are used to assess neurotransmitter levels, receptor densities, and hormonal status. For instance, assays can measure cortisol levels to assess HPA axis activity, thyroid hormones to rule out endocrine contributions, and inflammatory markers (e.g., C-reactive protein, cytokines) which are often elevated in depression [1, 2]. Brain-Derived Neurotrophic Factor (BDNF), a protein vital for neuronal growth and survival, is also frequently investigated, with lower levels often observed in depressed individuals [1].
Limitations and Clinical Translation: Despite these advanced methodologies, translating neurobiological findings into routine clinical practice for depression faces significant hurdles. Firstly, the heterogeneity of depression means that not all individuals exhibit the same neurobiological profile, making it difficult to establish universal diagnostic biomarkers [2]. Secondly, while research can identify correlations between brain changes and depressive symptoms, establishing definitive causality remains challenging. Furthermore, methodological limitations, such as variability in data acquisition protocols, preprocessing pipelines, and statistical analyses, can lead to inconsistent findings across studies [2]. Currently, neuroimaging and biomarkers are primarily used in research settings or to rule out other medical conditions, rather than for direct diagnosis of depression. The development of predictive models for treatment response, which integrate neurobiological data with clinical information using machine learning algorithms, shows promise for personalized medicine, but these models require extensive validation before widespread clinical adoption [3]. The ultimate goal is to move towards a more objective, neurobiologically informed approach to diagnosis and treatment selection, but this remains a future aspiration rather than current clinical reality [2, 3].
2. Environmental and Nutritional Pathogenesis
This section critically examines the intricate interplay between external environmental factors and nutritional status in the pathogenesis of depression. Moving beyond purely psychological explanations, it delves into the physiological pathways through which stressors such as air pollution, noise, and urban living conditions, alongside dietary patterns and deficiencies, can instigate or exacerbate depressive symptomatology. The focus is on understanding how these external influences perturb neurochemistry, trigger inflammatory responses, disrupt biological rhythms, and compromise neuronal integrity, thereby contributing to the complex etiology of depression.
2.1 Environmental Stressors and Neuroinflammation
The physical environment in which individuals live plays a significant, often underestimated, role in mental health, particularly in the development of depression. Modern urban environments, characterized by high levels of pollution, noise, and artificial light, present a constellation of stressors that can profoundly impact neurobiological processes. Exposure to fine particulate matter (PM2.5) in the air has been increasingly linked to an elevated risk of depression [4]. The proposed mechanisms for this association are multifaceted. Firstly, inhaled pollutants can trigger systemic inflammation, leading to the release of pro-inflammatory cytokines that can cross the blood-brain barrier or signal to the brain, thereby influencing mood regulation [4]. Secondly, these pollutants can induce oxidative stress by overwhelming the body's antioxidant defenses and generating reactive oxygen species (ROS). This oxidative damage can impair neuronal function, disrupt neurotransmitter synthesis, and contribute to neuronal dysfunction [4]. Chronic inflammation and oxidative stress are also implicated in impaired neurogenesis, particularly in the hippocampus, a brain region critical for mood regulation and stress response [4].
Noise pollution, a pervasive feature of urban life, acts as a chronic stressor, activating the sympathetic nervous system and leading to elevated cortisol levels. This sustained activation of the stress response system is a known contributor to mood disorders, including depression [4]. Furthermore, noise pollution significantly disrupts sleep quality and quantity. Poor sleep is a well-established risk factor for depression, exacerbating mood instability, cognitive deficits, and emotional dysregulation [4]. The constant auditory assault can also lead to cognitive impairment by interfering with attention and executive functions, further diminishing an individual's resilience to stress.
Artificial light at night (ALAN), or light pollution, disrupts the natural light-dark cycle, which is fundamental for regulating the body's circadian rhythms. This disruption can suppress melatonin production, a hormone crucial for sleep and mood regulation, and desynchronize the internal biological clock [4]. The consequences include sleep disturbances, hormonal imbalances, and alterations in the synthesis and release of key neurotransmitters such as serotonin and dopamine, all of which are implicated in the pathophysiology of depression [4].
Urban living conditions also encompass factors like limited access to green spaces and poor housing quality. A lack of exposure to nature is associated with higher rates of depression and anxiety, as green spaces provide opportunities for stress reduction, physical activity, and social interaction – all protective factors against depression [4]. The absence of these restorative environments limits their beneficial effects. Moreover, poor housing conditions, including overcrowding, dampness, inadequate ventilation, and insufficient natural light, contribute to chronic stress, feelings of helplessness, and can expose residents to environmental toxins like mold, which can trigger inflammatory responses [4]. These combined environmental stressors create a physiological milieu that is conducive to the development and maintenance of depressive symptoms, primarily through pathways involving neuroinflammation, oxidative stress, and circadian rhythm disruption.
2.2 Nutritional Deficiencies and Metabolic Pathways
The availability of essential nutrients is paramount for optimal brain function, including the synthesis and regulation of neurotransmitters, neuronal integrity, and neuroprotection. Deficiencies in key micronutrients and essential fatty acids can therefore significantly influence mood regulation and contribute to the development of depression. Vitamin D, often referred to as a prohormone, plays a critical role in brain health. Low levels of Vitamin D are frequently observed in individuals with depression, and it is hypothesized to influence mood through its presence in brain regions involved in mood regulation, its role in modulating the hypothalamic-pituitary-adrenal (HPA) axis, and its influence on serotonin synthesis [4]. Furthermore, Vitamin D possesses immunomodulatory and anti-inflammatory properties, which may counteract the pro-inflammatory state often associated with depression [4].
B vitamins, particularly B12, folate (B9), and B6, are indispensable cofactors in crucial metabolic pathways, most notably the methylation cycle. This cycle is vital for the synthesis of monoamine neurotransmitters such as serotonin, dopamine, and norepinephrine, which are central to mood regulation [4]. Deficiencies in these vitamins can impair neurotransmitter production and function. Elevated levels of homocysteine, a metabolic byproduct that can increase with B vitamin deficiencies, are also associated with an increased risk of depression and cardiovascular disease, suggesting a link through vascular and direct neurotoxic mechanisms [4].
Essential minerals also play critical roles. Magnesium is involved in NMDA receptor function and neurotransmitter release, and its deficiency can lead to increased neuronal excitability and heightened stress responses [4]. Zinc acts as a cofactor for numerous enzymes involved in neurotransmitter synthesis and is a component of antioxidant defense systems, protecting neurons from oxidative damage [4]. Iron is essential for the synthesis of dopamine and for adequate oxygen transport to the brain; iron deficiency anemia is well-known to cause fatigue and mood disturbances [4].
Omega-3 fatty acids, specifically eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), are integral components of neuronal cell membranes. They influence membrane fluidity, receptor function, and neurotransmitter signaling. Lower dietary intake and blood levels of omega-3s are associated with an increased risk of depression [4]. Their benefits are attributed to potent anti-inflammatory and antioxidant properties, which can mitigate the neuroinflammation and oxidative stress implicated in depression. Omega-3s may also promote neurogenesis and increase levels of brain-derived neurotrophic factor (BDNF), a protein crucial for neuronal survival, growth, and plasticity [4]. The evidence for dietary interventions and supplementation with these nutrients, while promising, is subject to ongoing research, with mixed results in some meta-analyses, highlighting the need for further investigation into optimal dosages, ratios, and specific patient populations [6].
2.3 The Gut-Brain Axis and Dietary Influence
The gut microbiome, the vast community of microorganisms residing in the gastrointestinal tract, has emerged as a critical regulator of brain function and mood through the bidirectional communication pathway known as the gut-brain axis. Dysbiosis, an imbalance in the composition and function of the gut microbiota, is increasingly linked to depression [4]. This axis operates through neural (e.g., the vagus nerve), endocrine, and immune pathways. Alterations in the gut microbiome can disrupt this communication, influencing neurotransmitter synthesis and signaling within the brain [4].
Gut bacteria are directly involved in producing or modulating the production of neurotransmitters and their precursors, such as serotonin, GABA, and dopamine. For instance, a significant portion of the body's serotonin is produced in the gut, and its synthesis is influenced by gut microbial activity [4]. Dysbiosis can lead to increased intestinal permeability, often referred to as 'leaky gut.' This allows bacterial products, such as lipopolysaccharides (LPS), to enter the bloodstream, triggering a systemic inflammatory response that can impact brain function and mood regulation [4].
Diet plays a pivotal role in shaping the gut microbiome and influencing systemic inflammation. Diets high in processed foods, refined sugars, and unhealthy fats are consistently associated with an increased risk of depression [4]. These dietary patterns promote systemic inflammation and oxidative stress due to their low fiber content, high glycemic load, and presence of pro-inflammatory compounds. They can negatively alter the gut microbiome, reducing the abundance of beneficial bacteria and increasing that of potentially pathogenic ones, leading to dysbiosis [4]. This dietary-induced dysbiosis can reduce the production of beneficial short-chain fatty acids (SCFAs), such as butyrate, which have anti-inflammatory and neuroprotective effects [4]. Conversely, diets rich in whole foods, fiber, fruits, and vegetables, such as the Mediterranean diet, support a diverse and healthy gut microbiome, reduce inflammation, and provide essential nutrients that support brain health and mood [6]. The evidence for dietary interventions like the Mediterranean diet suggests it can be a valuable strategy for both preventing and managing depressive symptoms, likely through its combined effects on the gut microbiome, inflammation, and nutrient provision [6]. The complexity of dietary patterns and the individual variability in gut microbiome composition present challenges in establishing definitive dietary prescriptions for depression, underscoring the need for personalized approaches and further research into specific food components and their mechanisms of action [6].
3. Organ-Related Etiologies and Systemic Impact
This section critically examines the intricate and often bidirectional relationship between organ dysfunction and the pathophysiology of depression. Moving beyond purely psychological explanations, it delves into the physiological mechanisms through which conditions affecting key organ systems—specifically the endocrine, gastrointestinal, and cardiovascular systems—can manifest as depressive symptoms. Furthermore, it explores how depression itself can precipitate or exacerbate pathology within these same organ systems, creating complex feedback loops that challenge conventional diagnostic and therapeutic approaches. The investigation focuses on how disruptions in hormonal regulation, gut-brain signaling, and cardiovascular health contribute to the neurobiological underpinnings of mood disorders.
3.1 Endocrine and Thyroid-Mediated Depression
The endocrine system, particularly the hypothalamic-pituitary-thyroid (HPT) axis, plays a pivotal role in regulating mood and energy metabolism. Dysfunction within this axis is a well-established contributor to depressive symptomatology. Hypothyroidism, characterized by reduced levels of thyroid hormones triiodothyronine (T3) and thyroxine (T4), directly impacts central nervous system function. These hormones are crucial for maintaining normal brain metabolism and are essential for the synthesis and regulation of key neurotransmitters, including serotonin and norepinephrine, both of which are implicated in mood regulation [7]. The physiological consequences of hypothyroidism can profoundly mimic the clinical presentation of major depressive disorder. Symptoms such as fatigue, lethargy, cognitive slowing, and a pervasive sense of sadness are directly attributable to the generalized metabolic slowdown induced by insufficient thyroid hormone levels. This phenomenon is clinically recognized as 'myxedema madness,' a term that underscores the profound psychological disturbances that can arise from severe hypothyroidism [7].
Conversely, the relationship is not unidirectional. Chronic stress and the physiological state of depression can also lead to adaptive changes in thyroid function. In conditions such as 'euthyroid sick syndrome' (also known as non-thyroidal illness syndrome), the body downregulates thyroid hormone production and metabolism as a protective mechanism in response to systemic inflammation or severe illness. In this state, circulating levels of T3 may be low, while TSH levels remain within the normal range. This adaptive response, while potentially beneficial in the short term for conserving energy during illness, can contribute to depressive symptoms by reducing the availability of active thyroid hormone in the brain [7]. The chronic nature of depression can thus lead to a persistent state of relative thyroid hormone deficiency, exacerbating mood disturbances. Furthermore, autoimmune thyroid diseases, such as Hashimoto's thyroiditis, are common causes of hypothyroidism and are often associated with mood disorders. The chronic inflammation and immune dysregulation inherent in these conditions can contribute to the complex interplay between the immune system, endocrine function, and mental health.
The HPT axis dysfunction in depression can be conceptualized as a disruption in the feedback mechanisms that regulate hormone release. In depression, there is often evidence of HPT axis hyperactivity, with elevated TSH levels in some individuals, suggesting impaired negative feedback from thyroid hormones. However, the picture is complex, with some studies showing blunted TSH responses to TRH (thyrotropin-releasing hormone) in certain subtypes of depression. This intricate dysregulation highlights how hormonal imbalances can directly contribute to the neurobiological changes associated with depression, affecting neurotransmitter systems, neuronal excitability, and energy metabolism within the brain.
3.2 Gastrointestinal Signaling and Microbial Metabolites
The gut-brain axis represents a sophisticated bidirectional communication network linking the central nervous system (CNS) and the gastrointestinal tract, heavily influenced by the gut microbiome. The gut itself functions as a significant endocrine and immune organ, and its microbial inhabitants play a critical role in modulating host physiology, including brain function and mood [8]. Dysbiosis, an imbalance in the composition and function of the gut microbiota, is increasingly recognized as a key factor in the pathophysiology of depression.
One primary mechanism through which gut dysbiosis impacts brain function is via the production of microbial metabolites. Short-Chain Fatty Acids (SCFAs), such as butyrate, propionate, and acetate, are produced by the bacterial fermentation of dietary fiber. Butyrate, in particular, has neuroprotective and anti-inflammatory properties; it acts as a histone deacetylase (HDAC) inhibitor, which can promote the expression of brain-derived neurotrophic factor (BDNF). BDNF is crucial for neuronal survival, growth, and synaptic plasticity, and its reduced levels are consistently observed in individuals with depression. A reduction in SCFA production due to dysbiosis can therefore impair synaptic plasticity and contribute to depressive-like behaviors [8].
Another critical pathway involves the metabolism of tryptophan, the essential amino acid precursor to serotonin. The gut microbiome significantly influences tryptophan availability for CNS serotonin synthesis. In states of dysbiosis and inflammation, the enzyme indoleamine 2,3-dioxygenase (IDO) becomes upregulated. IDO diverts tryptophan away from the serotonin synthesis pathway and towards the kynurenine pathway. This pathway produces various metabolites, including quinolinic acid and kynurenine. Quinolinic acid is a neurotoxic excitotoxin that acts as an agonist at NMDA receptors, contributing to neuroinflammation, oxidative stress, and neuronal damage. Kynurenine itself can also be neuroactive. The shift towards kynurenine pathway metabolites at the expense of serotonin production directly impairs mood regulation and promotes depressive states [8].
Immune-mediated pathways are also central to the gut-brain axis's role in depression. Dysbiosis can compromise the integrity of the intestinal epithelial barrier, leading to increased intestinal permeability, often referred to as 'leaky gut.' This allows bacterial products, such as lipopolysaccharides (LPS) from Gram-negative bacteria, to translocate from the gut lumen into the systemic circulation. LPS triggers a potent inflammatory response via toll-like receptor 4 (TLR4) on immune cells, leading to the release of pro-inflammatory cytokines like interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α). These cytokines can cross the blood-brain barrier or signal to the brain via circumventricular organs, activating microglia (the brain's resident immune cells) and inducing neuroinflammation. This neuroinflammatory state is a hallmark of depression and contributes to the 'sickness behavior'—a constellation of symptoms including fatigue, anhedonia, and social withdrawal—which shares significant phenotypic overlap with clinical depression [8].
Furthermore, certain gut bacteria can directly synthesize neurotransmitters such as gamma-aminobutyric acid (GABA) and serotonin. While these peripherally produced neurotransmitters may not readily cross the blood-brain barrier, they can act locally within the enteric nervous system (ENS) and communicate with the brain via the vagus nerve. This signaling can influence mood, stress responses, and gut motility [8]. The bidirectional nature of the gut-brain axis means that depression and chronic stress can also negatively impact gut health. Activation of the hypothalamic-pituitary-adrenal (HPA) axis and the release of stress hormones like cortisol can alter gastrointestinal motility, reduce mucosal blood flow, and modify the gut environment, creating conditions that favor dysbiosis [8]. This creates a vicious cycle where psychological distress exacerbates gut dysfunction, which in turn further contributes to depressive symptoms.
3.3 Cardiovascular and Immune System Interplay
The relationship between cardiovascular disease (CVD) and depression is robust and demonstrably bidirectional, mediated by a complex interplay of systemic inflammation, autonomic nervous system dysregulation, and shared risk factors. Depression is not merely a psychological reaction to having a cardiovascular condition; rather, it is an independent risk factor for the development and progression of CVD, including myocardial infarction (MI) and stroke [9]. Conversely, individuals with CVD are at a significantly elevated risk of developing depression.
A key unifying factor is chronic low-grade inflammation. Depression is increasingly understood as a state characterized by heightened systemic inflammation, evidenced by elevated levels of pro-inflammatory cytokines such as IL-6, TNF-α, and C-reactive protein (CRP). These inflammatory mediators contribute to endothelial dysfunction, a crucial early step in the development of atherosclerosis. Endothelial dysfunction impairs the ability of blood vessels to dilate properly, promotes the buildup of plaque, and increases the risk of blood clot formation. The chronic inflammatory milieu associated with depression physically accelerates the atherosclerotic process and can impair recovery following cardiovascular events [7, 9].
Autonomic nervous system (ANS) dysregulation is another critical link. In depression, there is often a shift in ANS balance towards sympathetic dominance and reduced parasympathetic (vagal) tone. This is reflected in decreased heart rate variability (HRV), which is a marker of impaired cardiovascular adaptability and a predictor of adverse cardiac events. Reduced HRV is associated with increased risk of arrhythmias, sudden cardiac death, and poorer outcomes after MI. Furthermore, sympathetic hyperactivation increases heart rate and blood pressure, contributing to hypertension and cardiac workload. It also promotes a pro-thrombotic state by increasing platelet reactivity, making blood clots more likely to form [7].
The immune system's role is central to this interplay. Depression can activate pro-inflammatory immune pathways, leading to a cascade of events that affect both the brain and the cardiovascular system. For instance, the activation of the kynurenine pathway, as discussed in the context of the gut-brain axis, also occurs in peripheral immune cells and contributes to systemic inflammation. This inflammation can directly impact cardiac tissue, contributing to myocardial dysfunction and increasing susceptibility to ischemic damage. The chronic stress response, often exacerbated in depression, further fuels this inflammatory state through the release of cortisol, which can have paradoxical pro-inflammatory effects in the context of chronic stress and lead to insulin resistance, a significant cardiovascular risk factor [7].
Conversely, cardiovascular disease itself can contribute to depression. Chronic hypoxia due to impaired cardiac function, reduced cerebral blood flow, and the physical limitations imposed by CVD can lead to psychological distress, anxiety, and depression. The burden of managing a chronic illness, the fear of future events, and the impact on quality of life all contribute to the high comorbidity rates. Moreover, inflammatory cytokines released from diseased cardiovascular tissue or activated immune cells can cross the blood-brain barrier, directly influencing neurochemistry and mood regulation [9].
In essence, depression can be viewed as a systemic illness that primes the body for cardiovascular pathology, while cardiovascular disease can create a biological and psychological environment conducive to depression. This creates a self-reinforcing feedback loop where each condition exacerbates the other. Therapeutic strategies that address both mental health and cardiovascular risk factors, such as regular exercise, stress management, and anti-inflammatory interventions, are crucial for breaking this cycle [9]. The shared pathways involving inflammation, autonomic dysfunction, and neuroendocrine dysregulation highlight the systemic nature of both depression and cardiovascular disease.
Conclusion and Future Directions
This research comprehensively investigated the multifaceted physical underpinnings of depression, examining its neurobiological correlates, the impact of environmental and nutritional factors, and the intricate relationship with organ system dysfunctions. Our review of neuroimaging and biomarker studies confirms that depression is not solely a psychological construct but is profoundly associated with tangible alterations in brain structure and function, including changes in the hippocampus, amygdala, prefrontal cortex, and anterior cingulate cortex. These neurobiological shifts are intrinsically linked to systemic physiological dysregulation, affecting the cardiovascular, immune, endocrine, and gastrointestinal systems, underscoring a complex, bidirectional interplay. Furthermore, this study synthesized evidence linking environmental stressors such as air and noise pollution, light pollution, lack of green space, and poor housing conditions, alongside nutritional factors like micronutrient deficiencies, inadequate omega-3 intake, high consumption of processed foods, and gut microbiome dysbiosis, to an increased risk and exacerbation of depressive symptoms. The investigation into organ-related etiologies revealed significant associations between thyroid dysfunction, gut-brain axis dysregulation, and cardiovascular health with depression, highlighting that these conditions can both precipitate and be exacerbated by depressive states.
The primary contribution of this research lies in its systematic synthesis of evidence demonstrating the pervasive physical nature of depression, moving beyond purely psychological interpretations. Methodologically, it highlights the utility of advanced neuroimaging techniques (fMRI, EEG, DTI), sophisticated computational models (GLM, ICA, machine learning), and biomarker analyses in delineating these complex relationships. Practically, the findings underscore the critical need for a holistic approach to depression management, integrating interventions that address neurobiological dysregulation, environmental exposures, nutritional status, and co-occurring organ pathologies. This research provides a foundation for developing more targeted and personalized therapeutic strategies.
Despite these advancements, significant limitations persist. A major constraint is the inherent difficulty in establishing definitive causal directions between neurobiological changes, organ dysfunction, environmental factors, and depression, given the prevalence of observational studies and the complex interplay of genetic, environmental, and lifestyle variables. The heterogeneity of depressive presentations further complicates the development of universal diagnostic and therapeutic models. Methodological challenges in accurately measuring environmental exposures and dietary intake, as well as the translational gap between research findings and routine clinical practice, also represent considerable hurdles. Consequently, the applicability of these conclusions is bounded by the current understanding of these complex multifactorial relationships and the limitations of existing research methodologies.
Looking forward, several critical research avenues warrant exploration. Future research should prioritize large-scale, longitudinal studies and randomized controlled trials to elucidate causal pathways and assess the efficacy of novel interventions. The development and validation of objective neurobiological and organ-specific biomarkers for depression diagnosis and treatment response prediction are paramount. Further investigation into the precise mechanisms of the gut-brain axis, the impact of environmental pollutant mixtures, and the role of other organ systems (e.g., liver, kidney) in depression is essential. Crucially, there is a pressing need to advance personalized medicine approaches, leveraging multi-omics data, advanced computational modeling, and AI to tailor interventions to individual neurobiological profiles, genetic predispositions, and specific organ system vulnerabilities. The ultimate goal is to transition towards a precision psychiatry model that can effectively prevent, diagnose, and treat depression based on its underlying physical pathology.
References
[1] llm_self_research
- Query: Neurobiological mechanisms of depression, including structural and functional brain changes, and their impact on specific organ systems and physiological processes.
- Summary: Depression is characterized by significant neurobiological alterations impacting brain structure and function, which in turn affect multiple organ systems and physiological processes. Structurally, key brain regions show modifications: the hippocampus often exhibits reduced volume, potentially impai...
[2] llm_self_research
- Query: Detailed methodologies and analytical techniques used to establish neurobiological correlates of depression and their impact on organ systems, including specific statistical models, neuroimaging analysis protocols (e.g., fMRI, EEG, DTI), and biomarkers. Also, explore current and potential future applications of this neurobiological understanding in clinical practice, such as diagnostic tools, therapeutic targets, and prognostic indicators for depression and associated organ dysfunction.
- Summary: This research explores the neurobiological underpinnings of depression and their systemic impact, detailing methodologies, analytical techniques, and clinical applications. Key brain regions implicated include the hippocampus (often reduced volume), amygdala (variable volume/activity), prefrontal co...
[3] llm_self_research
- Query: Explore methodologies and computational models used to analyze neuroimaging data (fMRI, EEG) in depression research, focusing on algorithms for identifying structural and functional brain changes, and investigate how neurobiological findings in depression are translated into diagnostic criteria, therapeutic targets, and predictive models for treatment response.
- Summary: This research explores the methodologies and computational models used in analyzing neuroimaging data (fMRI, EEG) for depression research, and investigates the translation of neurobiological findings into clinical applications.
Neuroimaging Methodologies and Computational Models:
fMRI Analysis:
...
[4] llm_self_research
- Query: Investigate the specific environmental factors (e.g., air pollution, noise pollution, light pollution, lack of green space, housing conditions) and nutritional factors (e.g., micronutrient deficiencies, omega-3 fatty acids, processed foods, gut microbiome) that have been scientifically linked to the development or exacerbation of depression. Focus on the proposed biological mechanisms and pathways involved, such as inflammation, oxidative stress, neurogenesis, and neurotransmitter function.
- Summary: This report synthesizes research on the environmental and nutritional factors linked to depression, focusing on proposed biological mechanisms.
Environmental Factors:
Air Pollution: Exposure to fine particulate matter (PM2.5) is correlated with an increased risk of depression. Proposed mechanisms...
[5] llm_self_research
- Query: Explore the methodologies and statistical models used in research investigating the link between environmental factors (air pollution, noise, light, green space, housing) and nutritional factors (micronutrients, omega-3, processed foods) with depression. Also, investigate practical applications of this knowledge in public health interventions, clinical practice, and policy-making, as well as current limitations and future research directions.
- Summary: This report details research methodologies, statistical models, and practical applications concerning the link between environmental and nutritional factors and depression. Methodologies employed include ecological, cohort, case-control, cross-sectional, longitudinal, and time-series studies, often ...
[6] llm_self_research
- Query: Explore specific dietary interventions for depression based on identified nutritional factors (e.g., Mediterranean diet, omega-3 supplementation, vitamin D supplementation) and their efficacy, including evidence-based guidelines and limitations. Additionally, research the methodologies for assessing the impact of environmental factors (e.g., air pollution, noise) on depression, including epidemiological study designs, biomarkers, and challenges in establishing causality. Finally, investigate the current limitations in research on environmental and nutritional factors in depression and suggest future research directions and potential interventions.
- Summary: This report synthesizes research on environmental and nutritional factors contributing to depression. Dietary interventions, including the Mediterranean diet, omega-3 supplementation, and vitamin D supplementation, show potential benefits. The Mediterranean diet, rich in fruits, vegetables, and heal...
[7] llm_self_research
- Query: Explore the hypothesis that organ dysfunctions or diseases can directly cause or significantly contribute to depression. Investigate physiological mechanisms linking conditions affecting organs like the thyroid, gut, and cardiovascular system to depressive symptoms. Also, examine whether depression can precipitate or worsen organ-specific pathology.
- Summary: Physiological Mechanisms of Organ-Depression Bidirectionality
Thyroid Dysfunction (The Endocrine Axis)
Mechanism: Hypothyroidism is strongly linked to depressive symptoms via the disruption of the hypothalamic-pituitary-thyroid (HPT) axis. Reduced levels of triiodothyronine (T3) and thyroxine (T4) ...
[8] llm_self_research
- Query: Explore the gut-brain axis mechanisms linking gut microbiome dysbiosis to depression, including specific microbial metabolites, immune pathways, and neurotransmitter modulation. Also, investigate the bidirectional relationship where depression may influence gut health and function.
- Summary: The Gut-Brain Axis: Mechanisms of Depressive Pathophysiology
Microbial Metabolites as Signaling Molecules
Short-Chain Fatty Acids (SCFAs): Microbial fermentation of dietary fiber produces butyrate, propionate, and acetate. Butyrate acts as a histone deacetylase (HDAC) inhibitor, promoting brain-der...
[9] llm_self_research
- Query: Explore diagnostic strategies and therapeutic interventions for depression that specifically consider organ-related etiologies, such as thyroid dysfunction and gut dysbiosis. Investigate the current limitations in understanding and treating these complex relationships, and identify emerging research directions or novel therapeutic approaches.
- Summary: This research explores diagnostic strategies and therapeutic interventions for depression linked to organ-related etiologies, specifically focusing on the thyroid and gut-brain axes. It highlights the bidirectional relationship between these organ systems and mood disorders, outlines current limitat...